 
Show that the following molecules and their corresponding shapes are correct, using VSEPR theory. Explain why the X-P-X bond angles for the series of POX 3 molecules decrease from X = Br (104.1°) to X = Cl (103.3°) to X = F (101.3°)įluorine is the most electronegative halogen, so it will draw electron density in the P-F bond away from P atom repulsion of the P-F bonding pairs will be less than the repulsion of P-Cl and P-Br bonding pairs, so the F-P-F bond angle will be the smallest.ĥ. Hence, H 2O will have the larger X-O-X bond angle.Ĥ. Which of H 2O and F 2O will have the larger X-O-X bond angle?į is more electronegative than H therefore the space occupied by the O-H bonding pair in the O valence shell will be greater. Total number of electron at the central I atom is: 7 + 2 -1 (charge) = 8 2BP + 2LP, should be tetrahedral and angular shape.ģ. Predict and draw the structure of I 3 + using VSEPR model. One Se-F and three Se-O (one terminal and two bridging) will add four more electrons to the valence shell of Se, so, 10 e = 5 electron pairs out of which four are bonded pairs and one is a lone pair. Draw the structure and depict the geometry around Se atoms in 3-, which is a symmetric ionic molecule with cyclic structure, using VSEPR model. Increasing the amount of p-character in the C-F bonds decreases the F-C-F bond angle, because for bonding by pure p-orbitals the bond angle would be decreased to 90°.ġ. The C-F bonds are formed from sp 3 + x hybrids, with slightly more p-character and less s-character than an sp³ hybrid, and the hydrogen are bonded by sp 3 - x hybrids, with slightly less p-character and slightly more s-character. While a carbon in CH 4 and CF 4 uses four identical sp³ hybrids for bonding, in CH 2F 2 the hybrids used are not identical. The bond angles in CH 4, CF 4 and CH 2F 2 can be explained using Bent’s rule. 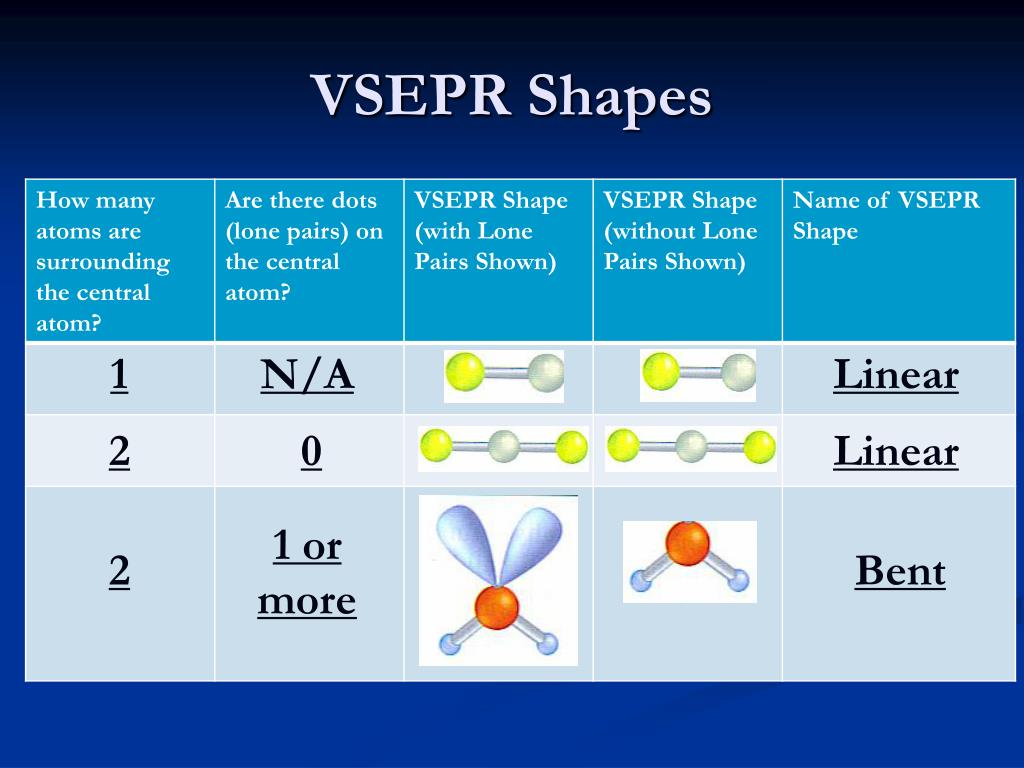 
More electronegative substituents ‘prefer’ hybrid orbitals having less s-character, and more electropositive substituents ‘prefer’ orbitals having more s-character. 
Multiple bonds count as a single bonding region. The shape of NH 3 molecule is pyramidal.Ĭount the number of bonding pairs and lone pairs around the central atom.Īrrange the bonding pairs and the lone pairs in one of the standard geometries thereby minimizing electron-electron repulsion. Similarly, in ammonia, the nitrogen atom is in tetrahedral environment with three bonded pairs (three N-H bonds) and one lone pair. The shape of the water molecule is therefore bent (two lone pairs are ignored). While mentioning the molecular shapes lone pairs may be ignored, however, while defining the geometry both the lone pairs and bond pairs should be considered.įor example: in water molecule the central oxygen atom is in tetrahedral environment with two lone pairs and two O-H bonds (or two bond pairs). Molecular shapes are eventually determined by two parameters: Bond distance, separation between the nuclei of two bonded atoms in a straight line and the bond angle, the angle between any two bonds containing a common atom. Since the lone pair-lone pair repulsions are maximum, the most stable geometry can be obtained by maximizing the distance between steric numbers on the central atom. \)Īnother term called steric number is often used in VSEPR theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
